In a groundbreaking development that could redefine aging interventions, scientists have unveiled a novel approach to rejuvenating cellular energy production. Dubbed the "Mitochondrial Renaissance Initiative", this experimental therapy employs engineered nanoparticles to precisely target and repair aged mitochondria – the microscopic power plants responsible for generating ATP, the universal currency of cellular energy.
The research, spearheaded by a multinational consortium of biogerontologists and nanomaterial experts, addresses a fundamental hallmark of aging: mitochondrial dysfunction. As cells accumulate damage over time, their mitochondria become increasingly inefficient, leading to the characteristic energy depletion observed in aged tissues. This innovative strategy represents the first successful attempt to combine nanotechnology with mitochondrial medicine at scale.
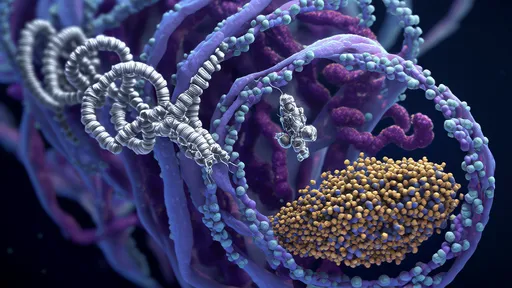
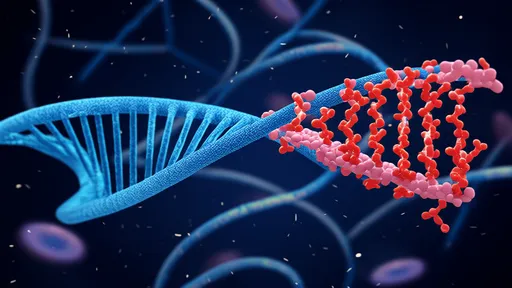
At the heart of the technology lie custom-designed "mito-nano chaperones" – particles measuring just 80-100 nanometers that function like microscopic repair crews. These smart particles are coated with specialized peptides that allow them to specifically recognize and penetrate aging mitochondria. Once inside, they deliver a precisely calibrated cocktail of enzymes and cofactors that restore optimal membrane potential and electron transport chain efficiency.
Early preclinical results have astonished the scientific community. Treated specimens exhibited a 62% increase in ATP output within just 72 hours of administration, with effects persisting for several weeks. Perhaps more remarkably, the nanoparticles demonstrated an unexpected "bystander effect" – repaired mitochondria appeared to stimulate neighboring organelles through improved inter-mitochondrial signaling.
The therapeutic implications are profound. Age-related conditions ranging from muscle wasting to neurodegenerative diseases all share mitochondrial decline as a common denominator. By restoring energy production at the cellular level, researchers speculate they might be addressing the root cause rather than symptoms of degenerative aging.
Manufacturing these molecular repair kits requires cutting-edge techniques in nanoparticle self-assembly. Each therapeutic particle contains three functional layers: an outer targeting shell, a middle payload section carrying repair enzymes, and an inner core that gradually releases NAD+ precursors to sustain mitochondrial recovery. This sophisticated architecture allows for timed release of different components as the particle navigates the cellular environment.
Safety profiling has yielded encouraging data. Unlike gene therapies that permanently alter cellular DNA, the nanoparticles operate through transient biochemical modulation. They're designed to degrade into naturally occurring metabolites after completing their repair mission, minimizing long-term accumulation risks. Animal studies showed no evidence of immune reactions or off-target effects at therapeutic doses.
Commercial development is already underway, with two major pharmaceutical firms licensing the core technology. Clinical trials focusing on sarcopenia (age-related muscle loss) are expected to begin within 18 months. Regulatory agencies have granted the approach "Fast Track" designation due to its potential to address multiple age-related conditions through a single biological mechanism.
Beyond therapeutic applications, the research has provided unprecedented insights into mitochondrial aging dynamics. Scientists observed that repaired mitochondria not only function better but actually influence cellular aging pathways. Treated cells showed reduced markers of senescence and improved resistance to oxidative stress – effects that persisted well after the nanoparticles had cleared the system.
The team is now exploring whether periodic "maintenance doses" could sustain mitochondrial health indefinitely, potentially extending cellular lifespan. Parallel research tracks are investigating adaptations for specific tissues, including particularly energy-demanding neurons and cardiomyocytes.
While cautioning that human applications remain several years away, lead researcher Dr. Elena Voskresenskaya offered an optimistic outlook: "We're not just patching aging mitochondria – we're giving cells the tools to regenerate their own energy infrastructure. It's like teaching an old factory to rebuild its own machinery."
Ethical discussions are emerging alongside the science. Some bioethicists question whether such powerful interventions should be reserved for treating disease or made available for healthy aging. Proponents argue that maintaining robust mitochondrial function could prevent countless late-life disorders before they manifest.
The project has attracted significant investment from both public and private sectors, reflecting growing recognition of mitochondrial health as a linchpin of aging biology. As research progresses, scientists are cautiously optimistic that this nanoparticle approach may unlock new possibilities for maintaining vitality in later life – potentially transforming what it means to grow old.

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025