In the intricate world of genome organization, a silent but fierce battle unfolds between molecular machines shaping our DNA. The protagonists—cohesin and CTCF—engage in a dynamic game that dictates how chromosomes fold, genes are regulated, and cellular identity is maintained. This high-stakes interaction, often dubbed the "topological wars," reveals a fascinating interplay between structure and function in the nucleus.
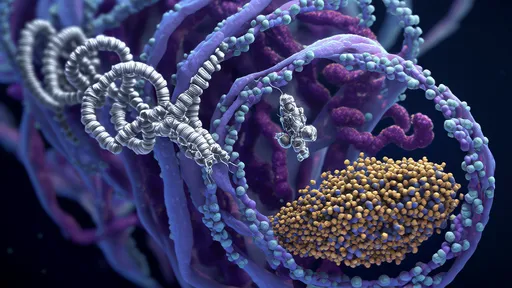
Cohesin, a ring-shaped protein complex, acts as a molecular motor that extrudes DNA loops with remarkable persistence. Like a relentless sculptor, it pulls chromatin fibers through its ring structure, creating ever-larger loops until meeting an unexpected adversary: CTCF. This zinc-finger protein, often called the "bookmark of the genome," halts cohesin’s advance by binding to specific DNA sequences. The resulting tension between these forces generates the complex 3D architecture of chromosomes observed across cell types.
The stakes of this confrontation couldn’t be higher. Proper DNA looping enables enhancers to find their target genes across vast genomic distances, while faulty topology can lead to developmental disorders or cancer. Recent live-cell imaging studies captured cohesin and CTCF in action, showing loops forming and dissolving within minutes—far more dynamically than previously assumed. This challenges the textbook view of static chromosomal territories and instead paints a picture of constant, energy-driven reorganization.
What makes this system particularly intriguing is its inherent asymmetry. While CTCF acts as a unidirectional barrier—stopping cohesin from one direction but allowing passage from another—the rules governing this polarity remain hotly debated. Some researchers propose that CTCF’s orientation depends on its binding motif’s sequence, while others suggest post-translational modifications create the directional signal. This molecular "one-way sign" ensures loops form productively rather than randomly, though the exact mechanism continues to elude scientists.
The conflict escalates during cellular transitions like differentiation or mitosis. As cells change identity, cohesin loading increases dramatically, flooding chromosomes with loop-extruding complexes. Meanwhile, CTCF binding sites appear and disappear from the genome like shifting battle lines. Single-cell techniques now reveal that identical genetic sequences can adopt strikingly different conformations in sister cells—proof that topological heterogeneity contributes to cellular plasticity. Such findings blur the line between genetic determinism and three-dimensional chance.
Cancer genomes expose the consequences of this delicate balance breaking down. Mutations disrupting CTCF binding or cohesin function frequently appear in malignancies, causing enhancers to activate wrong genes or structural variations to accumulate. Therapies targeting these components—such as cohesin inhibitors in acute myeloid leukemia—are now entering clinical trials, turning fundamental discoveries into potential treatments. Yet the full therapeutic implications remain uncertain, given these proteins’ roles in all DNA-templated processes.
Emerging evidence suggests the battlefield extends beyond simple loop formation. Cohesin and CTCF may collaborate in certain contexts, working together to create specialized chromatin compartments. Super-resolution microscopy reveals transient "handshakes" between the two proteins, hinting at unappreciated cooperation amidst their famous opposition. Some loops even appear stabilized by additional factors like WAPL or PDS5, adding layers of regulation to the basic extrusion-barrier model.
The evolutionary perspective adds another dimension to this story. While cohesin’s core structure remains conserved from yeast to humans, CTCF gained new functions in vertebrates—particularly at imprinted loci and X-chromosome inactivation centers. This suggests the topological wars intensified alongside developmental complexity, perhaps enabling sophisticated gene regulation required for multicellular life. Even viruses pirate this system, with Epstein-Barr virus positioning its genome near CTCF sites to hijack host loops.
As CRISPR-based screens identify new players in this field and computational models better predict loop dynamics, one truth becomes clear: The dance between cohesin and CTCF represents more than mere structural biology. It’s a living, breathing system that translates linear genetic information into four-dimensional function—with implications spanning from basic development to precision medicine. The topological wars rage on, and with each skirmish, we gain deeper insight into the choreography of life itself.

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025
By /Aug 18, 2025
By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025

By /Aug 18, 2025